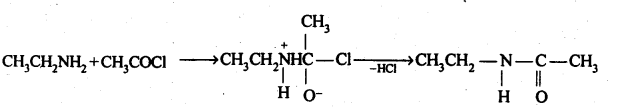 
For further information on the types of chemical reactions compounds such as acetyl chloride can undergo, see acyl halide. Acetyl is an acyl group having the formula-C(=O)-CH3. The smoke is actually small droplets of hydrochloric acid and acetic acid formed by hydrolysis.Īcetyl chloride is used for acetylation reactions, i.e., the introduction of an acetyl group. In fact, if handled in open air it releases white "smoke" resulting from hydrolysis due to the moisture in the air. It also arises from the reaction of acetic acid, acetonitrile, and hydrogen chloride.Īcetyl chloride is not expected to exist in nature, because contact with water would hydrolyze it into acetic acid and hydrogen chloride.  
It can also be synthesized from the catalytic carbonylation of methyl chloride. When heated, a mixture of dichloroacetyl chloride and acetic acid gives acetyl chloride. However, these methods usually give acetyl chloride contaminated by phosphorus or sulfur impurities, which may interfere with the organic reactions. It is a colorless, corrosive, volatile liquid.Īcetyl chloride is produced in the laboratory by the reaction of acetic acid with chlorodehydrating agents such as PCl3, PCl5, SO2Cl2, phosgene, or SOCl2. It belongs to the class of organic compounds called acyl halides. Acetyl chloride (CH3COCl) is an acid chloride derived from acetic acid.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
